 Number of Protons + Number of Neutrons ATOMIC MASS / WEIGHTĦ NEUTRONS- I need to know the number of neutrons….ĭo you know the atomic mass / weight AND the number of protons? If so, subtract the number of protons from the atomic mass/ weight. In each case, enough information has been provided for you to fill in the blanks. Do you know the number of protons and neutrons? If so, add the number of protons and neutrons to get the atomic mass / weight. Part A Subatomic Particles The table below contains information about several elements. Question 1 Shown here is a simplified representation of an atom: the smallest division of matter that may be isolated through physical or chemical methods. Do you know the number of protons? If so, then they are the same number.ĥ ATOMIC MASS / WEIGHT- I need to know the atomic mass / weight…ĭo you have a periodic table of elements? If so, find the largest number (number with the greatest value) and round it to the nearest whole number, if necessary. Choose 1 answer: Protons, neutrons, and photons A Protons, neutrons, and photons Positrons, neutrons, and electrons B Positrons, neutrons, and electrons Protons, electrons, and positrons C Protons, electrons, and positrons Protons, neutrons, and electrons D Protons, neutrons, and electrons Stuck Review related articles/videos or use a hint. ATOM NUCLEUS The nucleus is the center of mass (A), butdoes not significantly contribute to volume.It is made up of: PROTONS: Mass 1 amu, charge +1 NEUTRONS: Mass 1 amu, charge 0 ELECTRONS The electronic cloud determines the size, or volume,ntribute of tothe mass atom, but does not significantlyELECTRONS: Mass. Do you know the number of electrons? If so, then they are the same number. Do you know the number of protons? If so, then they are the same number!Ĥ ATOMIC NUMBER- I need to know the atomic number….ĭo you have a periodic table of elements? If so, then find the number the periodic is organized by, and that is the atomic number.  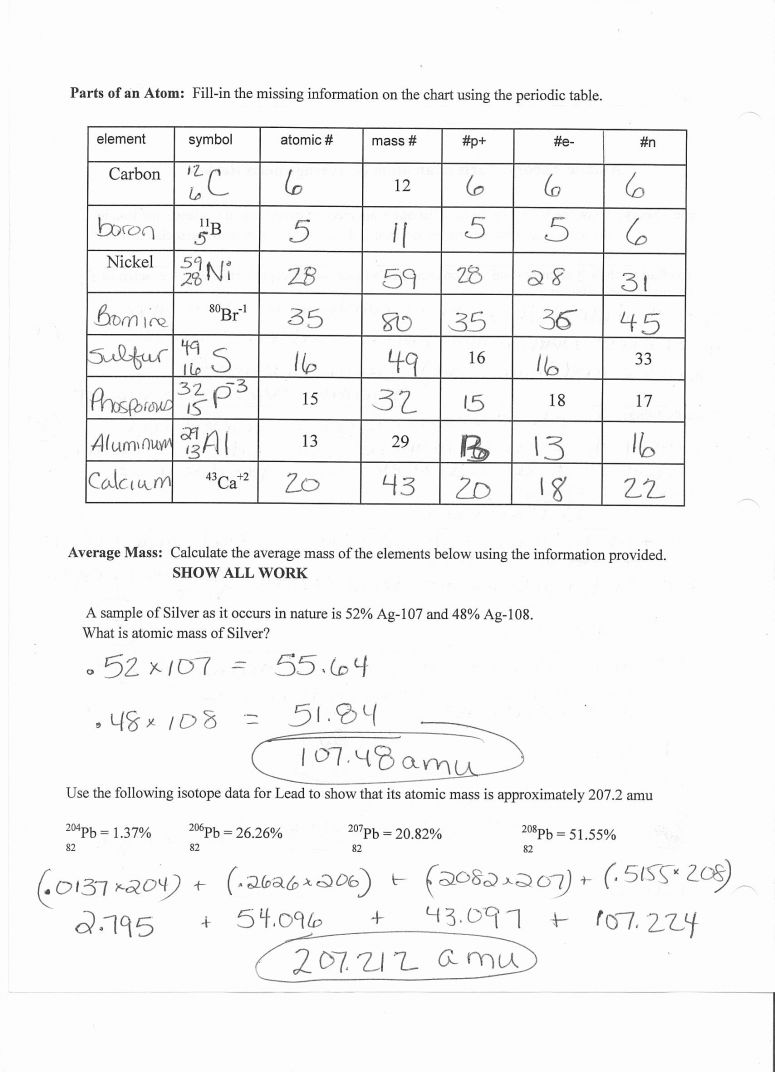
Do you know the number of electrons? If so, then they are the same number.ģ ELECTRONS- I need to know the number of electrons….ĭo you know the atomic number? If so, then they are the same number. 
Do you know the atomic number? If so, then they are the same number. 2 PROTONS- I need to know the number of protons….ĭo you have a periodic table of elements? If so, the number of protons is the same as the atomic number, the smaller number. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
